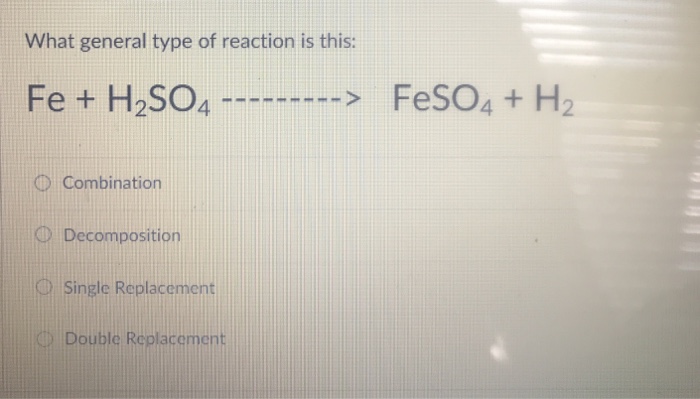
balance the following chemical equation by providing the correct coefficients fe+h2so4 fe(so4)3 + h2 help - brainly.com

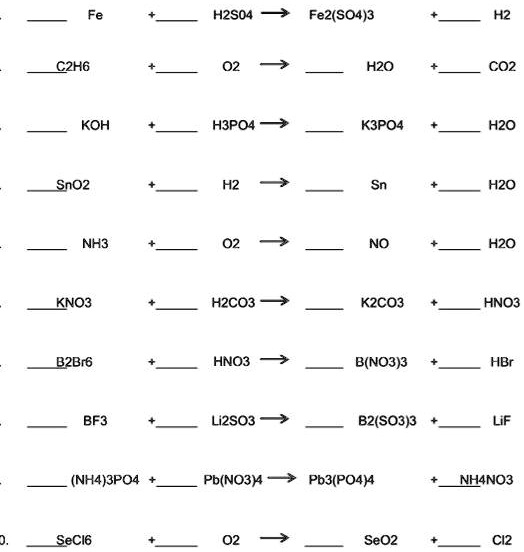
1. Fe + H2SO4Fe2(SO4)3 + H22. C2H2 + O2H2O + CO23. KOH + H3PO4K3PO4 + H2O4. SnO2 + H2Sn + H2O5. NH3 + 02NO + - Brainly.in

How to Balance Fe + H2SO4 = FeSO4 + Fe2(SO4)3 + H2O + SO2 (Iron + Concentrated Sulfuric acid) - YouTube

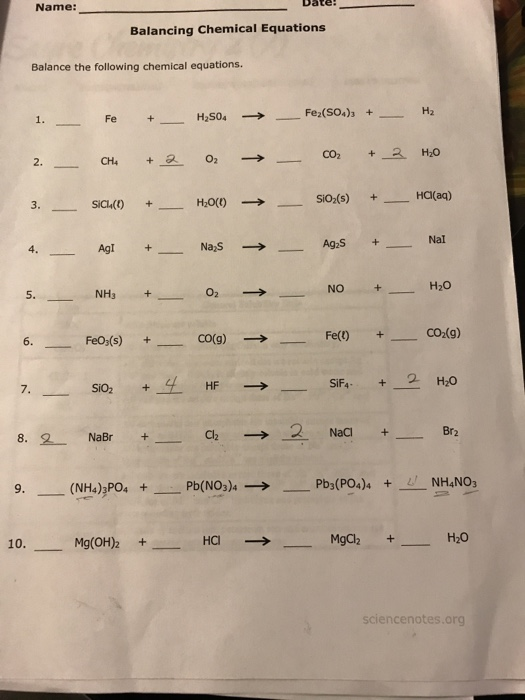
Balancing Equations 1 A.pdf - Name: Date: Balancing Equations Balance The Following Chemical - CHEM181 | Course Hero

Fe(OH)3 + H2SO4 = Fe2(SO4)3 + H2O does anyone know the net ionic equation for this rxn? : r/chemhelp

Question Video: Identifying the Balanced Symbolic Equation for the Reaction of Iron Metal with Concentrated Sulfuric Acid | Nagwa

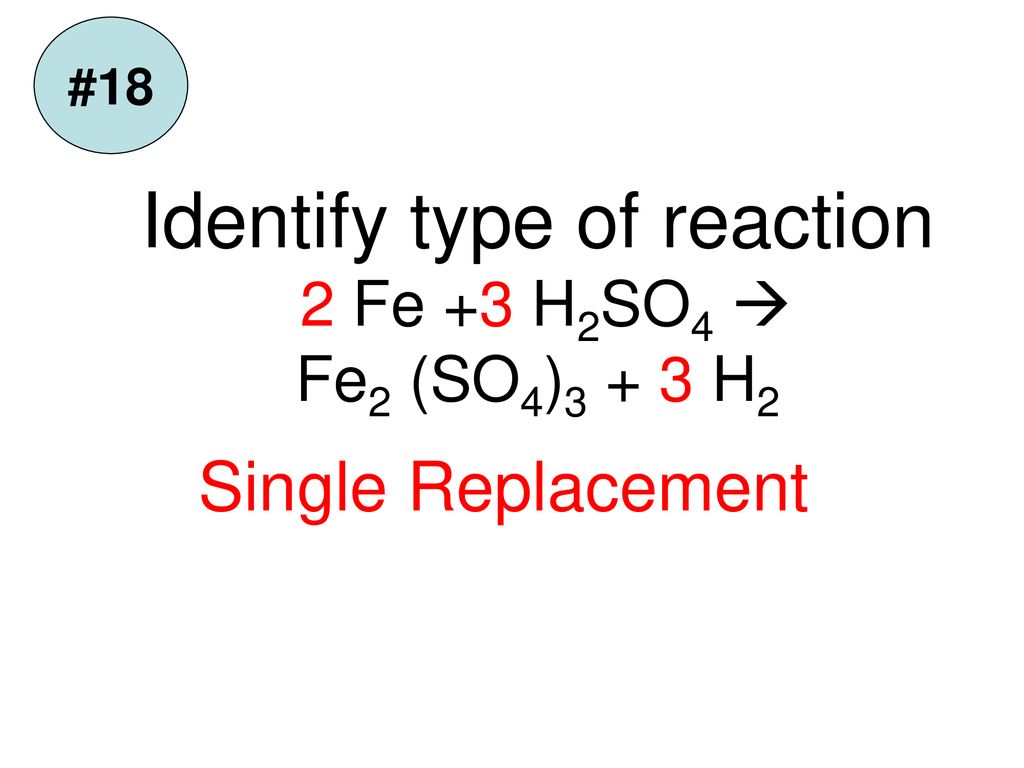
5. Which one of the following reaction is not possible (a) Fe + H2SO4 → FeSO4+H2 (b) Cu + 2AgNO3 → Cu (NO3)2 + 2Ag (c) 2KBr + 12 +2KI + Brz (d) CuO+H2 → Cu + H20

The Oxidation of Fe(II) in Acidic Sulfate Solutions with Air at Elevated Pressures. Part 1. Kinetics above 1 M H2SO4 | Industrial & Engineering Chemistry Research









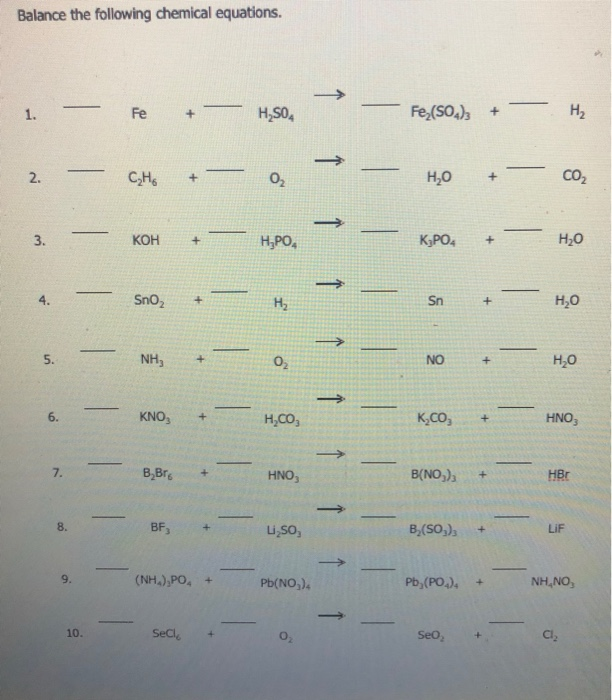
![middle school: balancing equations] How do i balance these equations? : r/chemistryhomework middle school: balancing equations] How do i balance these equations? : r/chemistryhomework](https://i.redd.it/e53006xcr2s51.jpg)